JOURNAL 1252
Journal of Chemical Metrology
Year: 2019 Issue: 1 January-June
p.21 - 28
Viewed 3852 times.
GRAPHICAL ABSTRACT
ABSTRACT
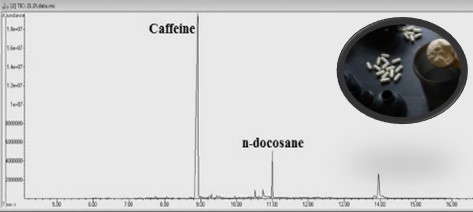
Caffeine is a substance that can be easily consumed in daily nutrition as well as in sport supplements. Exceeding the daily-recommended dose for caffeine may cause serious health problems. Studies illustrated that many concerns have been raising about the incompatibility between labels on sport supplements and their contents. The present study aimed to investigate caffeine contents in sport supplements purchased from Turkey market for the first time and to evaluate label information on products. Caffeine concentrations were determined in 41 sport supplements and possible adverse effects were emphasized considering calculated daily consumption doses. Seventeen samples among 41 products were found to be caffeine containing, ranging from 4.52 to 471 mg/mL. Three of caffeine containing products were undeclared their caffeine ingredients. Although three products were not specified caffeine their amounts, findings were found above the daily-recommended caffeine limit (400 mg/day). Significant differences were observed between the quantities indicated on the labels and the determined caffeine amounts in 7 products (p<0.05). Eight of 17 caffeine labelled products were found between 151-948% higher than stated label information. Overall, 29.4% of caffeine containing products (n=17) were found over the recommended intake. Consuming more than the daily-recommended dose may cause severe adverse reactions and consumers may experience serious health risks such as arterial hypertension, coronary heart disease, as well as sleep problems, irritability, anxiety, headache, and restlessness. Considering these health risks, awareness of consumers and accurate product labels on sport supplements gain more importance to prevent people from undesired consequences.
KEYWORDS- Sport supplement
- caffeine
- GC-MS
- label inconsistency
- daily intake