JOURNAL 2139
Organic Communications
VOLUME & ISSUE
Year: 2021 Issue: 3 July-September
Year: 2021 Issue: 3 July-September
PAGES
p.294 - 299
p.294 - 299
STATISTICS
Viewed 2696 times.
Viewed 2696 times.
GRAPHICAL ABSTRACT
ABSTRACT
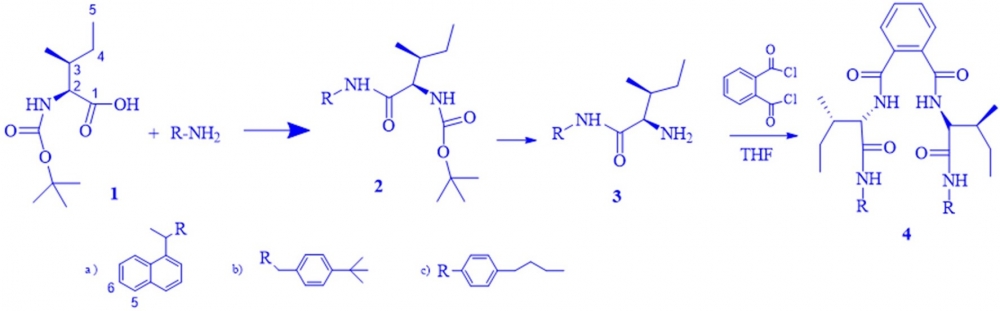
N,N′-Dicyclohexylcarbodiimide and hydroxybenzotriazole mediated amination of free COOH group of N-tert-butyloxyoxycarbonyl-L-isoleucine 1 with three aromatic amines gave the corresponding amides 2a-c. Deprotection of N-tert- butyloxyoxycarbonyl groups of 2a-c with AcOH yielded the related amine derivatives 3a-c. The amidation of the free NH2 groups of (3a-c) with phthaloyl dichloride yielded the bis-amide products 4a-c. The synthesized compounds were purified by crystallization and their structures were elucidated by spectroscopic methods such as 1H-HMR, 13C-NMR, FTIR, and micro analysis.
KEYWORDS- Amides
- tetraamides
- N-boc-deprotection
- coupling reaction
- spectroscopy
- heterocyclic