JOURNAL 3778
Records of Natural Products
VOLUME & ISSUE
Year: 2026 Issue: 3
Year: 2026 Issue: 3
PAGES
p.7 - 7
p.7 - 7
STATISTICS
Viewed 13 times.
Viewed 13 times.
AUTHORS
-
Hao Fan

-
Xin Wang

-
Yinhan Teng

-
Pingping Wu

-
Haifang Wang

-
Yuze Li

-
Henglei Niu

-
Wei Wang

-
Xiaomei Song

-
Qing Chang

-
Dongdong Zhang

GRAPHICAL ABSTRACT
ABSTRACT
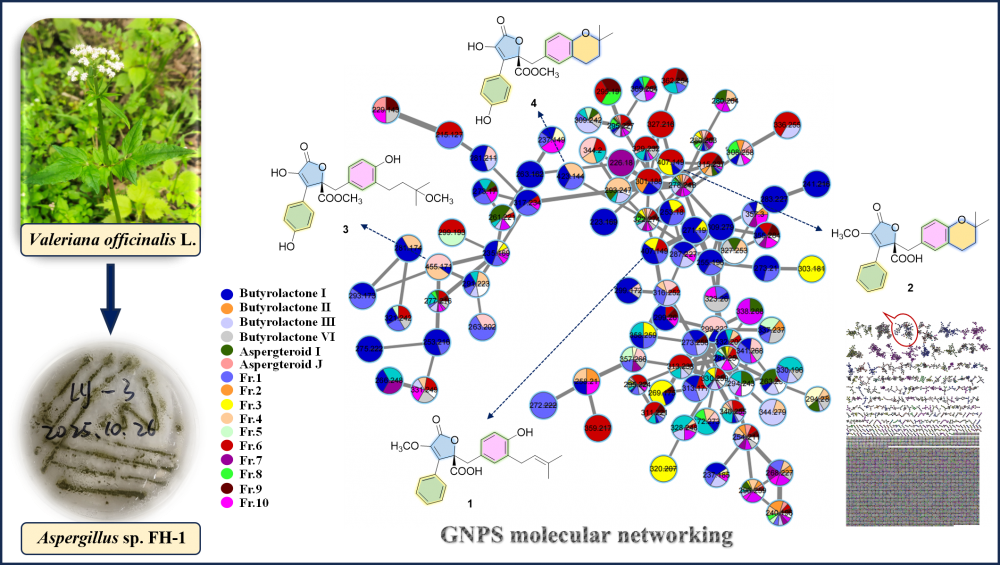
Guided by an integrated GNPS molecular networking strategy, the chemical investigation of endophytic fungus Aspergillus sp. FH-1 (isolated from Valeriana officinalis L.) led to the discovery of four butenolides (1-4). Their structures were elucidated through comprehensive spectroscopic data analysis, including HRESIMS, 1D/2D NMR and ECD, revealing two new congeners, asperianas A (1) and B (2), along with the known analogues 3 and 4. Notably, compounds 1 and 3 exhibited significant antifungal activity against Colletotrichum gloeosporioides, with EC50 values of 19.23 ± 1.14 and 43.36 ± 1.12 μg/mL, respectively.
KEYWORDS- Aspergillus sp. FH-1
- GNPS molecular networking
- butenolides
- antifungal