Records of Natural Products
A scientific open access journal in the field of natural products.LATEST ARTICLES
GNPS molecular networking-guided discovery of antifungal butenolides from the endophytic fungus Aspergillus sp. FH-1 of Valeriana officinalis L.
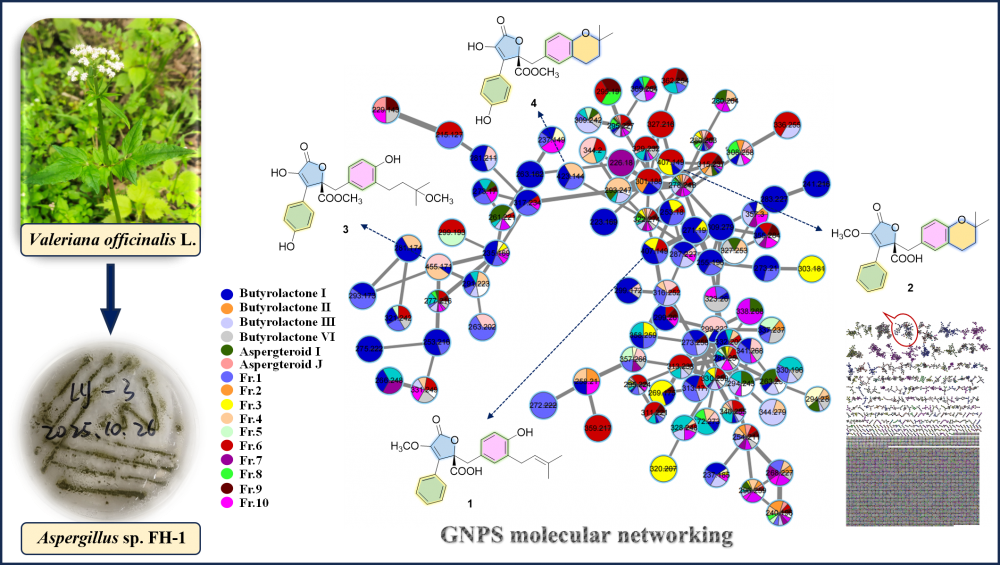
Guided by an integrated GNPS molecular networking strategy, the chemical investigation of endophytic fungus Aspergillus sp. FH-1 (isolated from Valeriana officinalis L.) led to the discovery of four butenolides (1-4). Their structures were elucidated through comprehensive spectroscopic data analysis, including HRESIMS, 1D/2D NMR and ECD, revealing two new congeners, asperianas A (1) and B (2), along with the known analogues 3 and 4. Notably, compounds 1 and 3 exhibited significant antifungal activity against Colletotrichum gloeosporioides, with EC50 values of 19.23 ± 1.14 and 43.36 ± 1.12 μg/mL, respectively.
DOI http://doi.org/10.25135/rnp.2512.3778 Keywords Aspergillus sp. FH-1 GNPS molecular networking butenolides antifungal DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.Two new cucurbitane-type triterpenoids fromthe peel of Trichosanthes kirilowii

Two new cucurbitane-type triterpenoids (1-2) and one known compound (3) were obtained from the ethyl acetate extract of Trichosanthes kirilowii Maxim. The structures of these compounds were elucidated on the basis of spectroscopic data analysis and comparison with spectroscopic data in the literature. All compounds were evaluated for anti-inflammatory activity in LPS-induced RAW264.7 cells. Compounds 1 and 2 could inhibit the secretion of IL-6, TNF-α, and NO, with IC50 values ranging from 34.82±1.54 to 86.14±1.94 μM
DOI http://doi.org/10.25135/rnp.2512.3776 Keywords Trichosanthes kirilowii triterpenoids anti-inflammatory DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.An experimental and in silico study on the antifungal activity of guaianolides isolated from Centaurea polypodiifolia Boiss. against resistant Candida strains
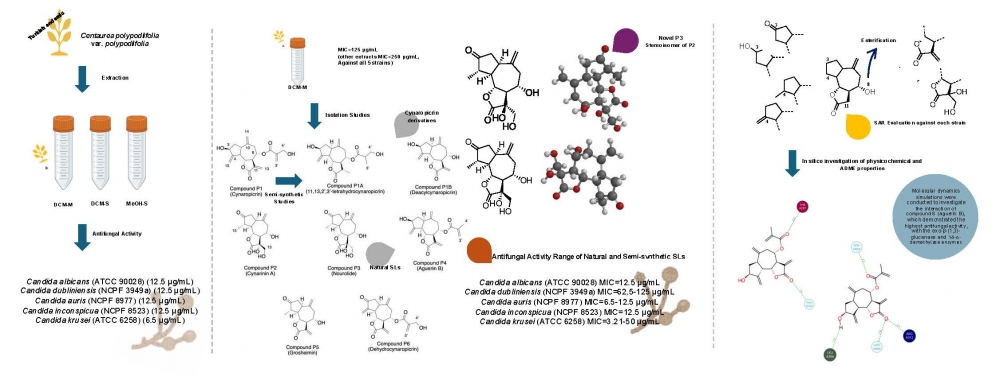
The emergence of multidrug-resistant Candida strains especially, Candida auris, C. dubliniensis, and C. inconspicua has accelerated the research on antifungal novel molecules. Sesquiterpene lactones, abundant in the Asteraceae family, have drawn attention with their antifungal activity. Therefore, three extracts obtained from the aerial part of Turkish endemic Centaurea polypodiifolia were investigated against Candida albicans (ATCC 90028), C. krusei (ATCC 6258), C. dubliniensis (NCPF 3949a), C. inconspicua (NCPF 8523), and C. auris (NCPF 8977) and gave MIC values between 6.25 and 12.5 µg/mL. Dichloromethane (DCM-M) extract yielded five known (cynaropicrin, cynarinin A, aguerin B, grosheimin, and dehydrocynaropicrin) and one novel (nourolide) guaianolide-type sesquiterpene lactones. Two semi-synthetic cynaropicrin derivatives were obtained to enhance the molecular diversity of the present study. 1D (1H and 13C APT) and 2D (COSY, HSQC, HMBC, and NOESY) NMR experiments were employed for structure elucidation. MIC values of the tested sesquiterpene lactones against the above-mentioned strains ranged between 3.12 and 50 µg/mL. Molecular dynamics simulations were conducted to investigate the interaction of 4 (aguerin B), which demonstrated the highest antifungal activity, with the exo-β-(1,3)-glucanase and 14-α-demethylase enzymes. These findings revealed stable binding interactions, suggesting that aguerin B has potential as a lead for further antifungal drug development.
DOI http://doi.org/10.25135/rnp.2512.3763 Keywords antifungal Candida Centaurea sesquiterpene lactone fungal resistance DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.New polyketide and chromene derivative isolated froman endophytic Diaporthe Phaseolorum associated with Polygonatum cyrtonema Hua
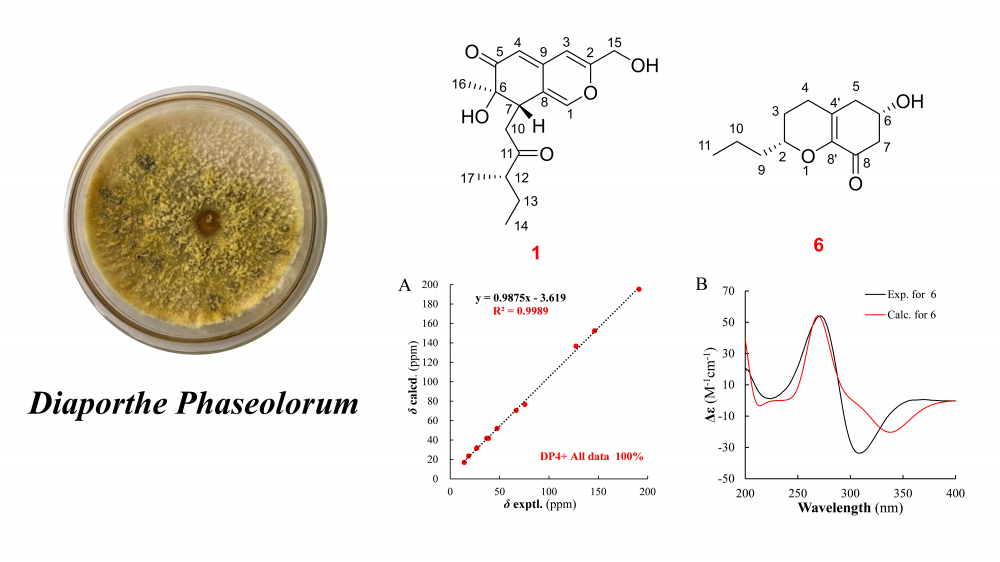
Eight compounds were isolated from the ethyl acetate extract of the Diaporthe phaseolorum fermentation products, comprising five polyketide derivatives (1–5), one chromene (6), one chromone (7), and one tetralone (8). Among these, compounds 1 and 6 were identified as previously undescribed. Notably, compound 6 features a unique C12 chromone skeleton, representing only the second reported example of such a framework. The structures and spatial configurations of all compounds were elucidated through comprehensive analysis of HR-MS, NMR, and quantum chemical calculations supported by DP4+ analysis. In vitro anti-inflammatory activity screening revealed that none of the isolated compounds (1–8) inhibited nitric oxide (NO) production in mononuclear macrophages.
DOI http://doi.org/10.25135/rnp.2601.3781 Keywords Diaporthe phaseolorum Polygonatum cyrtonema Hua polyketide chromene DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.
