Records of Natural Products
A scientific open access journal in the field of natural products.LATEST ARTICLES
Chemodiversity studies on Cleome L. genera by chemical characterization of Cleome pallida Kotschy and C. fimbriata Vicary essential oils with subsequent hierarchical cluster (HCA) and principal component analysis (PCA)
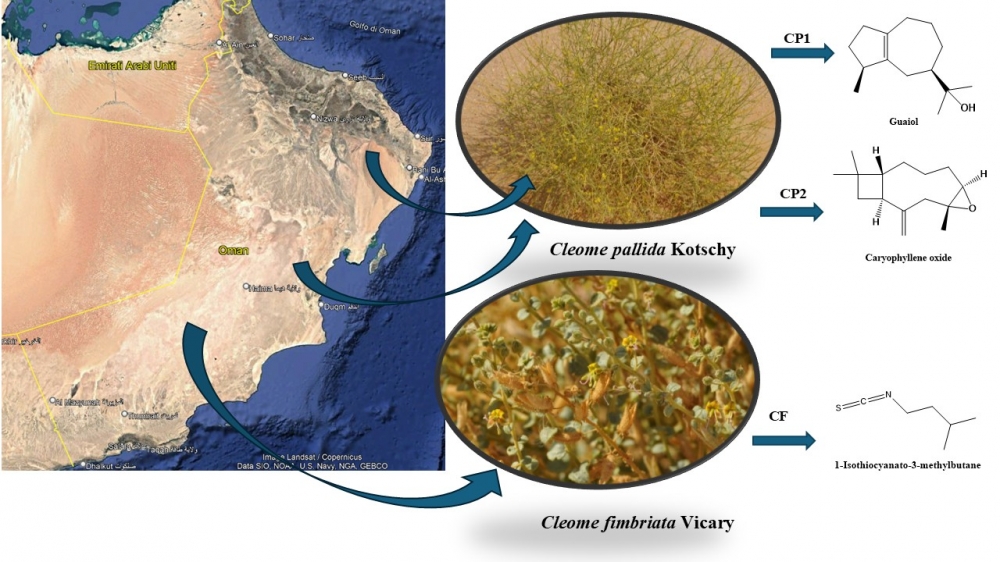
Cleome genus, belonging to the Cleomaceae family, includes about 180-199 species distributed in several countries and many of which are used extensively in ethnomedicine. In fact, many of these specimens are consumed in the form of infusions or salads to treat multiple physiological problems. In this study, the qualitative chemical composition of the essential oils obtained by hydrodistillation from Cleome ssp. was studied. Two species, two different accessions (CP1, and CP2) of C. pallida Kotschy, and one of C. fimbriata Vicary (CF), were collected in Oman and chemically investigated for the first time. The three essential oils, extracted by hydrodistillation, were analyzed by gas chromatography and mass spectrometry (GC-MS). In total, 169 compounds were identified. Sesquiterpenoids were found to be the major chemical group of CP1 and CP2 samples (56.4-58.8%); CF, instead, consisted essentially of nitrogen derivatives (30.2%), with 1-isothiocyanato-3-methylbutane (20.7%) as the most abundant class’ compound. Furthermore, a new complete review of the chemical compositions’ literature, never studied, was carried out on all other Cleome species. spontaneous in the world, studied so far. Statistical studies such as Hierarchical cluster (HCA) and principal component analysis (PCA) were employed used to highlightestablish chemo-similarities or dissimilarities, and to highpoint possible correlations between the chemical compositions of CP1, CP2, and CF, and the pharmacognosy and the reported Cleome ssp. essential oils.
DOI http://doi.org/10.25135/rnp.2601.3788 Keywords Caryophyllene oxide ecological traits food properties 1-isothiocyanato-3-methylbutane volatile analyses DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.Tenuifoliside Z1, an undescribed glycolipid from Polygala tenuifolia Willd. roots with antioxidant activity
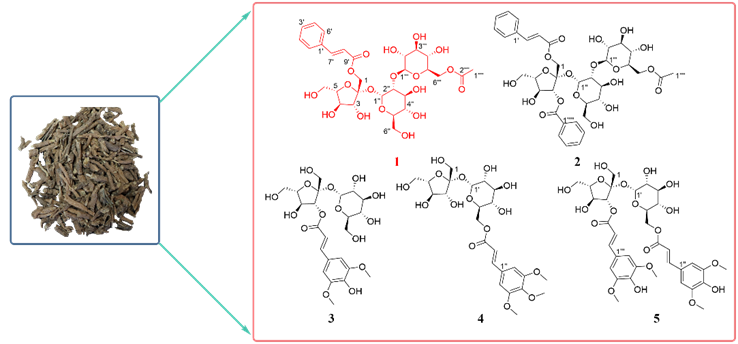
Abstract: a new glycolipid, tenuifoliside Z1 (1), together with four known glycolipid compounds (2-5) has been isolated from the root of Polygala tenuifolia Willd. Their structures were determined through spectroscopic analysis, chemical derivatization, and comparison with spectroscopic data reported in the literature. All the compounds were evaluated for antioxidant activity by in vitro assays.
DOI http://doi.org/10.25135/rnp.2601.3791 Keywords Polygala tenuifolia Willd Polygalaceae Glycolipids Antioxidant activity DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.Anti-inflammatory xanthones from the fruits of Hypericum patulum Thunb.
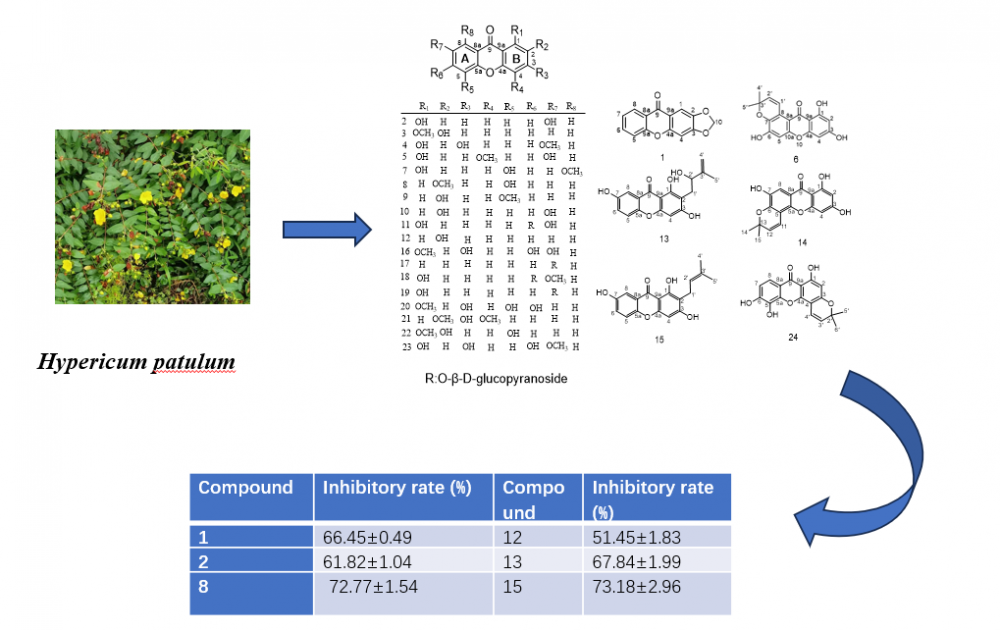
Abstract: Twenty-four known xanthones were isolated from the 80 % ethanol extract of the fresh ripe fruits of Hypericum patulum Thunb. Their structures were elucidated by extensive NMR and MS spectroscopic analysis and comparison with literature data. Compounds 4, 7, 14, and 18-19 were first reported in this genus. Compounds 1, 3, 9-11, 13, 15-17, and 20-21 were first reported from this species. Anti-inflammatory studies have shown that compounds 1-2, 8, 12-13 and 15 have a significant inhibition rate of NO release compared to dexamethasone (DEX). The anti-inflammatory activity data formed the basis for the structure-activity relationship analysis of the isolated compounds.
DOI http://doi.org/10.25135/rnp.2512.3760 Keywords Hypericum patulum Thunb. Xanthones anti-inflammatory activity structure-activity relationship chemotaxonomy DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.GNPS molecular networking-guided discovery of antifungal butenolides from the endophytic fungus Aspergillus sp. FH-1 of Valeriana officinalis L.
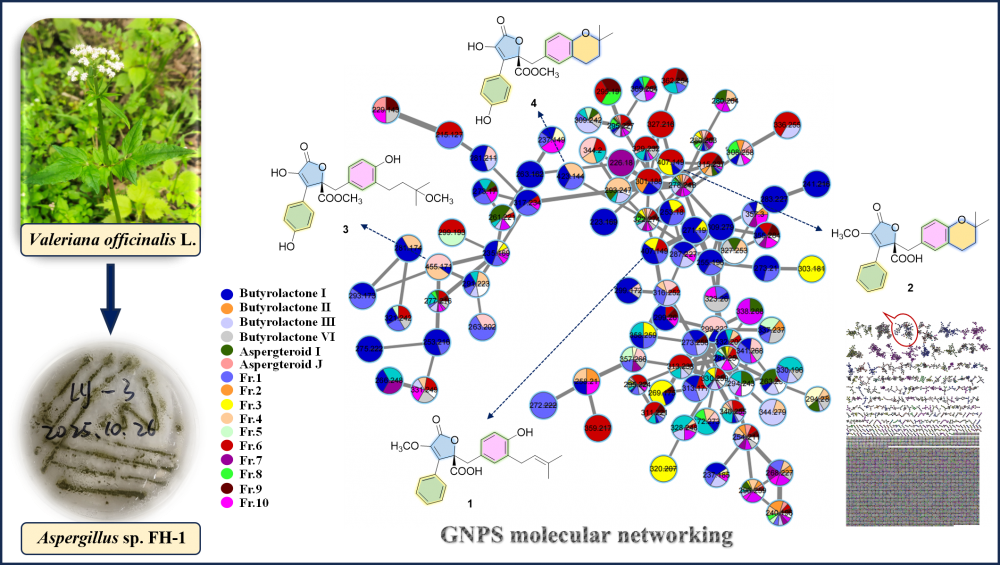
Guided by an integrated GNPS molecular networking strategy, the chemical investigation of endophytic fungus Aspergillus sp. FH-1 (isolated from Valeriana officinalis L.) led to the discovery of four butenolides (1-4). Their structures were elucidated through comprehensive spectroscopic data analysis, including HRESIMS, 1D/2D NMR and ECD, revealing two new congeners, asperianas A (1) and B (2), along with the known analogues 3 and 4. Notably, compounds 1 and 3 exhibited significant antifungal activity against Colletotrichum gloeosporioides, with EC50 values of 19.23 ± 1.14 and 43.36 ± 1.12 μg/mL, respectively.
DOI http://doi.org/10.25135/rnp.2512.3778 Keywords Aspergillus sp. FH-1 GNPS molecular networking butenolides antifungal DETAILS PDF OF ARTICLE © 2026 ACG Publications. All rights reserved.
